Selective effector cells agonist for IO
This is illustrated by AU-007, the first-ever computationally designed antibody to enter human clinical trials. AU-007 was designed by Biolojic to precisely bind to pre-defined epitope on interleukin-2 (IL-2) to redirect IL-2 toward cells that express dimeric receptors (T effs, NKs, NKTs), and away from cells that express the trimeric receptor (T regs, vascular endothelium). Thus, AU-007 unlocks the full potential of IL-2 to fight cancer: it starves the cells that protect the tumor and nourishes that cells that kill the tumor.
Unlocking the Power of IL-2
Interleukin-2 is often described as a “double-edged sword” because of its ability both to suppress and activate the immune system. For cancer patients, increased levels of IL-2 correlate with improved survival.
Dual Specific Antibody Targeting Two Cytokines
Our anti-IL-13 / TSLP multibody is designed to treat inflammatory diseases. This is the first multibody ever in development.
Current drugs that attack this pathway are blockbusters, validating the great need, but each of them works only for about half of the patients. Why?
The pathway of these diseases involves changing levels of the thymic stromal lymphopoietin (TSLP) and of Interleukin 13 (IL-13) over time. Existing drugs focus on only one of these two drivers of inflammation, and do not respond to pathway dynamics.
BD9 can block both TSLP and IL-13. When TSLP is upregulated BD9 binds it with both arms and ultra-high affinity. When IL-13 is upregulated, BD9 can bind it with both arm and very high affinity. Thus, BD9 can help more patients and improve outcomes for existing respondents.
This is an example of how Biolojic’s technology can solve one of the greatest challenges in drug development: BD9 will be given to all patients in the same way and yet it will have personalized activity – it will adjust its activity to what is needed for a specific patient, at a specific time in a specific tissue.
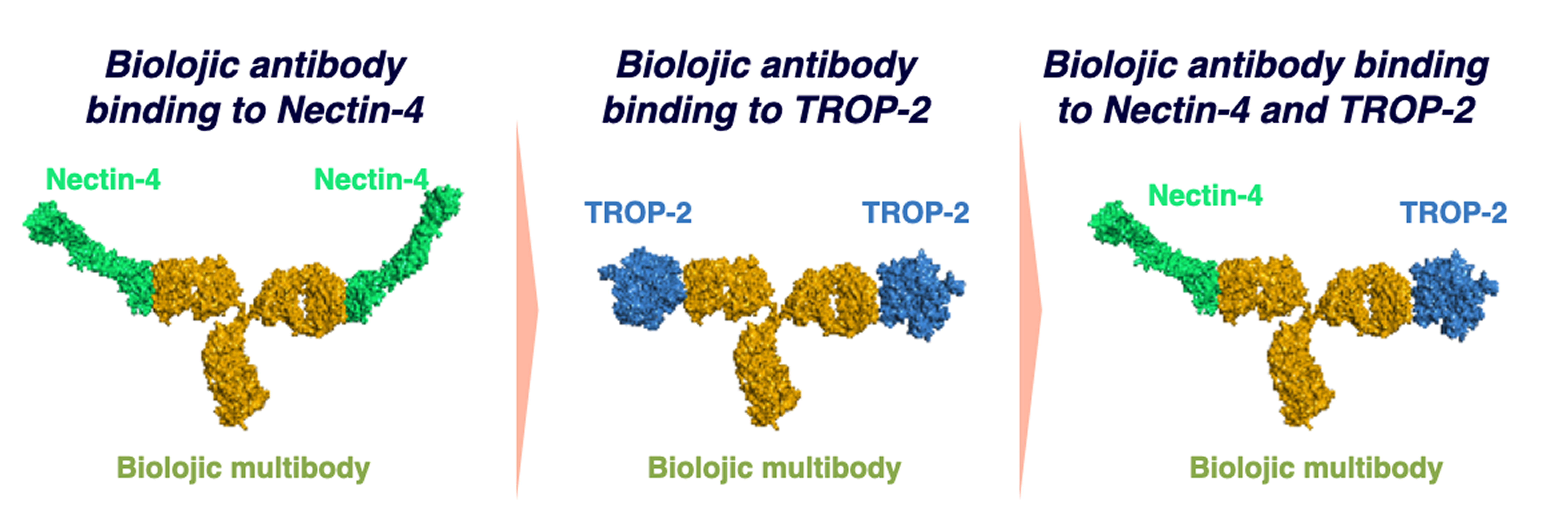
Dual binding Multibody Anti-Nectin-4 /TROP-2 : Base structure for an ADC
- Targeting two TAAs with overlapping expression profiles on tumor cells to strengthen the attack
- Avoiding down regulation of antigens as escape mechanisms
- Mitigating against heterogeneous expression of a single tumor antigen
- Broadening span of relevant indications – indications expressing either one of the TAAs or the two
- Symmetrical IgG construct (not a bispecific)
- Uniquely suited to overcome target expression heterogeneity – both arms can bind both targets