Unlike conventional drugs, which behave the same way everywhere, these programmable antibodies can sense the micro-environment in a specific patient at a specific time and respond dynamically.
Designing Programmable Antibodies
When your immune system encounters an antigen, it searches for an existing antibody (in the immune memory or in the germline) that will serve as a template to create a new antibody for that specific antigen. Next, your immune system begins making specific mutations to that template antibody, creating a new antibody that will bind to the new target.
We take a similar approach, using artificial intelligence (AI). Over the past decade, we’ve collected billions of measurements on billions of antibodies. This rich proprietary dataset informs computer algorithms that help design antibodies with new capabilities. In each project, we generate billions of new data points that allow us to train and refine dedicated machine learning models specifically for the task at hand.

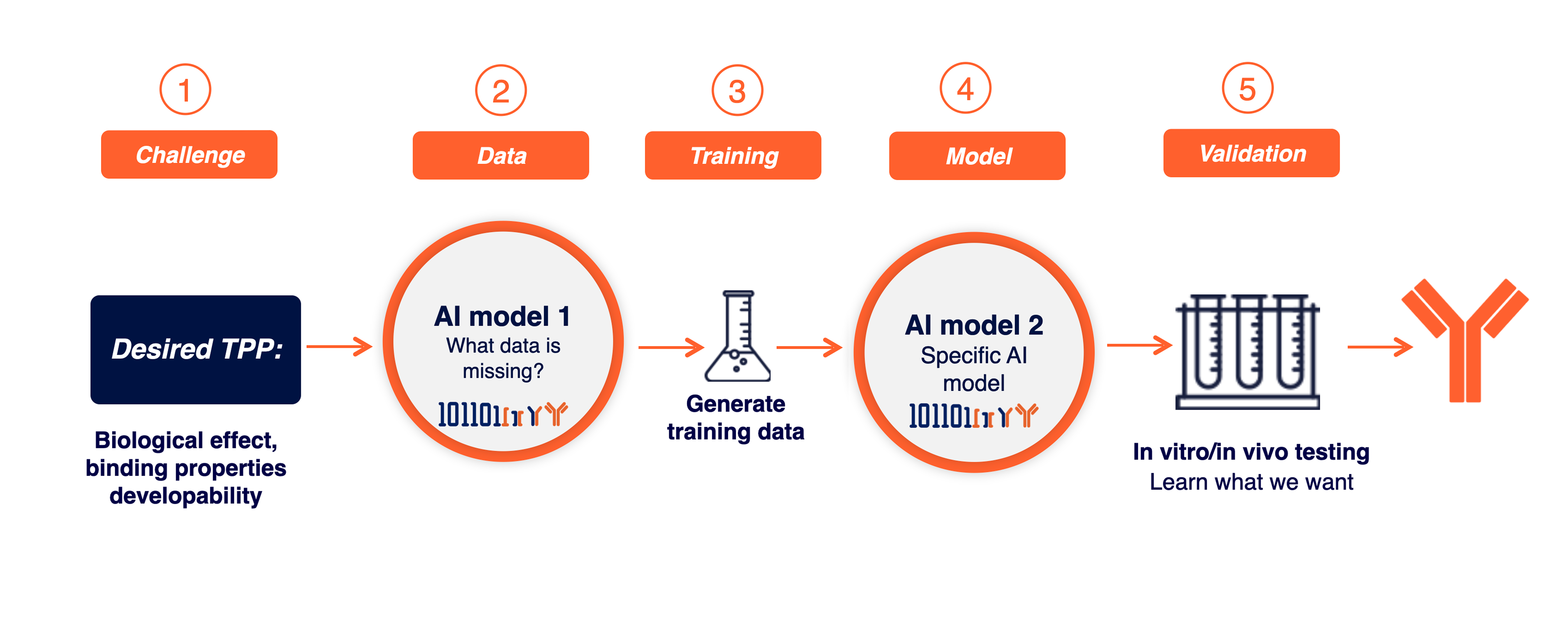
- Input – Desired TPP
- First AI model identifies a template antibody that can serve as the starting point and suggests specific mutations that will cause the selected template antibodies to bind the desired target(s) with the desired functional effect
- High throughput experiments generate data on the effect of specific mutations on antibody function and character
- Second AI model is trained on the new experimental data
- Output – A sequence of an optimized developable antibody that’s meets the TPP